Battery construction
The word battery simply means a group
of similar components. In military vocabulary, a "battery"
refers to a cluster of guns. In electricity, a "battery" is
a set of voltaic cells designed to provide greater voltage
and/or current than is possible with one cell alone.
The symbol for a cell is very simple,
consisting of one long line and one short line, parallel to
each other, with connecting wires:
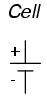
The symbol for a battery is nothing more
than a couple of cell symbols stacked in series:
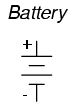
As was stated before, the voltage produced
by any particular kind of cell is determined strictly by the
chemistry of that cell type. The size of the cell is
irrelevant to its voltage. To obtain greater voltage than
the output of a single cell, multiple cells must be
connected in series. The total voltage of a battery is the
sum of all cell voltages. A typical automotive lead-acid
battery has six cells, for a nominal voltage output of 6 x
2.2 or 13.2 volts:
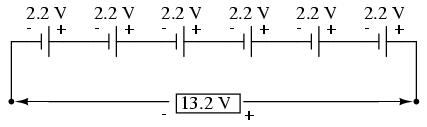
The cells in an automotive battery are
contained within the same hard rubber housing, connected
together with thick, lead bars instead of wires. The
electrodes and electrolyte solutions for each cell are
contained in separate, partitioned sections of the battery
case. In large batteries, the electrodes commonly take the
shape of thin metal grids or plates, and are often referred
to as plates instead of electrodes.
For the sake of convenience, battery symbols
are usually limited to four lines, alternating long/short,
although the real battery it represents may have many more
cells than that. On occasion, however, you might come across
a symbol for a battery with unusually high voltage,
intentionally drawn with extra lines. The lines, of course,
are representative of the individual cell plates:

If the physical size of a cell has no impact
on its voltage, then what does it affect? The answer is
resistance, which in turn affects the maximum amount of
current that a cell can provide. Every voltaic cell contains
some amount of internal resistance due to the electrodes and
the electrolyte. The larger a cell is constructed, the
greater the electrode contact area with the electrolyte, and
thus the less internal resistance it will have.
Although we generally consider a cell or
battery in a circuit to be a perfect source of voltage
(absolutely constant), the current through it dictated
solely by the external resistance of the circuit to
which it is attached, this is not entirely true in real
life. Since every cell or battery contains some internal
resistance, that resistance must affect the current in any
given circuit:
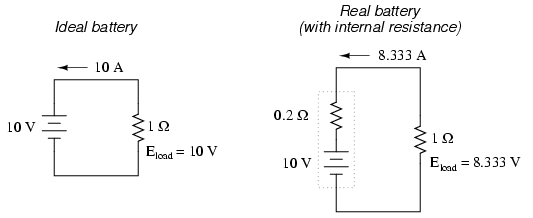
The real battery shown above within the
dotted lines has an internal resistance of 0.2 Ω, which
affects its ability to supply current to the load resistance
of 1 Ω. The ideal battery on the left has no internal
resistance, and so our Ohm's Law calculations for current
(I=E/R) give us a perfect value of 10 amps for current with
the 1 ohm load and 10 volt supply. The real battery, with
its built-in resistance further impeding the flow of
electrons, can only supply 8.333 amps to the same resistance
load.
The ideal battery, in a short circuit with 0
Ω resistance, would be able to supply an infinite amount of
current. The real battery, on the other hand, can only
supply 50 amps (10 volts / 0.2 Ω) to a short circuit of 0 Ω
resistance, due to its internal resistance. The chemical
reaction inside the cell may still be providing exactly 10
volts, but voltage is dropped across that internal
resistance as electrons flow through the battery, which
reduces the amount of voltage available at the battery
terminals to the load.
Since we live in an imperfect world, with
imperfect batteries, we need to understand the implications
of factors such as internal resistance. Typically, batteries
are placed in applications where their internal resistance
is negligible compared to that of the circuit load (where
their short-circuit current far exceeds their usual load
current), and so the performance is very close to that of an
ideal voltage source.
If we need to construct a battery with lower
resistance than what one cell can provide (for greater
current capacity), we will have to connect the cells
together in parallel:
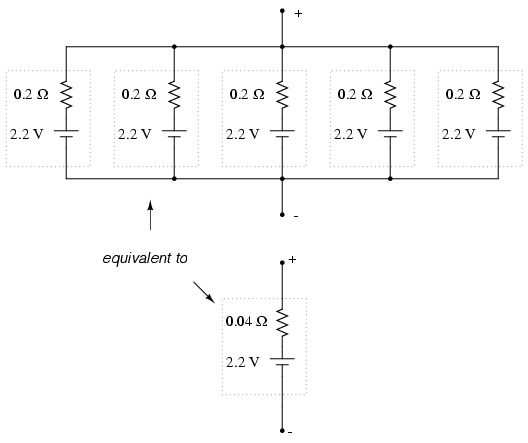
Essentially, what we have done here is
determine the Thevenin equivalent of the five cells in
parallel (an equivalent network of one voltage source and
one series resistance). The equivalent network has the same
source voltage but a fraction of the resistance of any
individual cell in the original network. The overall effect
of connecting cells in parallel is to decrease the
equivalent internal resistance, just as resistors in
parallel diminish in total resistance. The equivalent
internal resistance of this battery of 5 cells is 1/5 that
of each individual cell. The overall voltage stays the same:
2.2 volts. If this battery of cells were powering a circuit,
the current through each cell would be 1/5 of the total
circuit current, due to the equal split of current through
equal-resistance parallel branches.
-
REVIEW:
-
A battery is a cluster of cells
connected together for greater voltage and/or current
capacity.
-
Cells connected together in series
(polarities aiding) results in greater total voltage.
-
Physical cell size impacts cell
resistance, which in turn impacts the ability for the cell
to supply current to a circuit. Generally, the larger the
cell, the less its internal resistance.
-
Cells connected together in parallel
results in less total resistance, and potentially greater
total current.
|